לאתר שלנו בעברית, אנא הקישו כאן

Lab Members :

Vikas Dwivedi PhD, Post Doc

Sela Yechezkel, Agronomist

Sarah Sharf, Volunteer
Former lab members:
Maoz Lahav, MSc student , finished 2004
Shelly Zaffriar, MSc student, finished 2005
Baruch Zimerman, Post Doc, finished 2006
Lior Golomb, MSc student, finished 2007
Michal Halpert, MSc student finished 2008
Dr. Dror Avisar, Post Doc, finished 2010
David Szwerdszarf, finished 2010
Dr. Yoni Maskovitch, Post Doc, finished 2011
Osnat Altshuler, Msc student, finished 2013
Oxana Rogovoy, Post Doc, finished 2012
Aviv Levy, MSc student, finished 2013
Raj Kumar Thapa, MSc student, finished 2013
Inna Mordehaev, Bsc student, finished 2013
Marina Grumberg, MSc student, finished 2014
Saar Leninkron, BSc student, finished 2014
Ori Serero, MSc student, finished 2018
Avi Eliyahu, MSc student, finished 2019
Sapir Hagai, BSc student, finished 2019
Zvi Duman, Msc student, finished 2020
Mohamad Abu-Abied, finished 2020
Gal Hadas-Brandwein, Msc student, finished 2022
Pann Tzeela, Mac student, finished 2022
Lab Interests:
The focus of the lab is on the following subjects:
Plant Cell Biology and the Cytoskeleton
Plant myosins
Plant myosins belong to two main classes of unconventional myosins: myosin XI and myosin VIII, both closely related to myosin V. The Arabidopsis thaliana myosin family comprises 17 members: 13 myosin XI variants (myosin XI-A-K, XI-1 and XI-2) and 4 myosin VIII variants (ATM1, ATM2, myosin VIII-A and VIII-B). It is well-established that myosin XI family members propel the motility of large organelles, small vesicles, the endoplasmic reticulum (ER) and the whole cytoplasm. Nevertheless, the mechanism underling the process is not fully understood. The main confusing observation is that while motility of Golgi stacks, peroxisomes and mitochondria is reduced in myosin xik-knockout mutant plants, a functional full length myosin XIK fussed to YFP does not localize to these organelles. Three mechanisms were proposed to explain this puzzle: 1. Myosin XI bind specific transport vesicles that move along actin fibers and create a hydrodynamic flow of the cytoplasm which is the cytoplasmic streaming. All other organelles and vesicles are transported by this flow without a direct interaction with myosin XI. 2. Myosin XI binds directly to vesicles but very transiently and propel only short runs along actin tracks, thus very difficult to capture. 3. The amount of myosin molecules binding simultaneously an organelle is low and under detectable level.
We study the possibility that spatial and temporal post translational modifications of myosin XI tail regulate its binding to different organelles.
Interestingly, functional myosin XIK-YFP expressed in the xik/xi2/xi1 triple mutant was found in the spindle and phragmoplast mid zone in dividing cells both in root and shoot apical meristems, suggesting additional function in cell division. This is also a subject in our research.
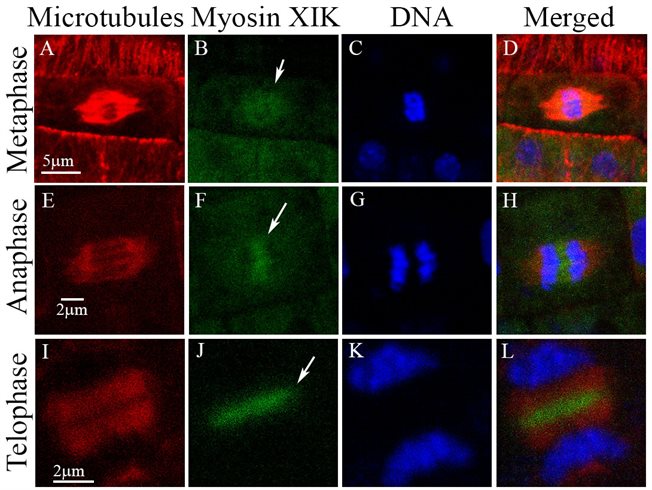
Myosin XIK in the cell division apparatus (Abu-Abied et al. 2018)Plant microtubules
Plant microtubules
The plant microtubules (MTs) cytoskeleton is organized into two main structures: during cell division, it forms the cell-division apparatus, which is sequentially developed from the preprophase band, to the spindle and then the phragmoplast; during interphase, MTs form parallel arrays under the plasma membrane, termed cortical MTs. Cortical MTs remain tethered to the plasma membrane along their entire length during interphase. Under the microscope, unlabeled cortical MTs are sometimes seen as black slits in the distribution pattern of labeled membrane associated proteins. According to one explanation, this happens because cortical MTs can restrict membrane protein lateral diffusion in the plasma membrane by forming "fences". We showed that cortical MTs can also collide with membrane associated proteins and change their orientation accordingly.
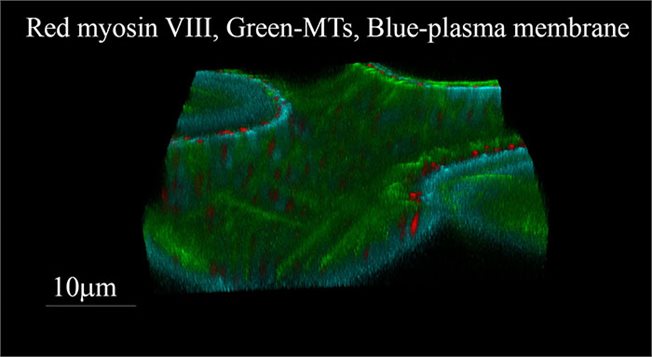
Three dimensional view of epidermal cell of Nicotiana benthamiana expressing markers for MTs (GFP) and for ATM1 tail (RFP).
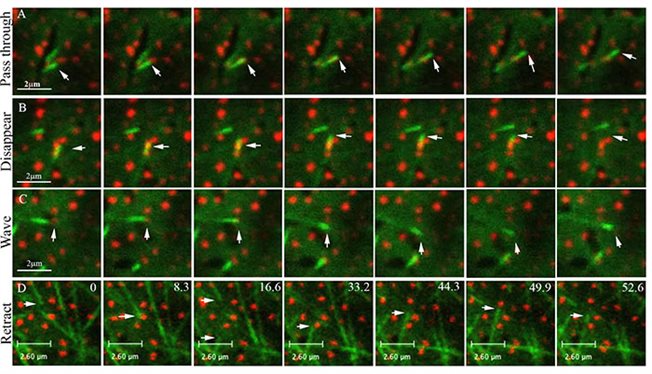
Different scenarios of interaction between elongated ends of microtubules marked by EB1-YFP and the tail of the myosin VIII membrane protein marked by RFP (Bar-Sinai et al. 2022)
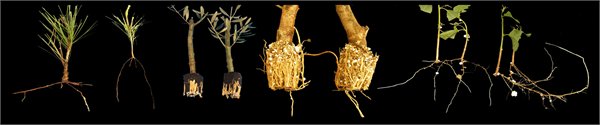
Molecular mechanisms underlying Adventitious Root Formation
Molecular mechanisms underlying Adventitious Root Formation
Adventitious roots are formed from non-root tissues. Root induction from stem cuttings is one of the common ways for vegetative propagation and is an important agricultural practice. Rooting of cuttings is economically important in forestry, in the ornamental plant industry and in development of elite rootstocks to increase yield, provide resistance to pests, diseases and environmental conditions. Despite the major economic and agricultural importance of vegetative propagation, a major barrier still exists for propagating clones of valuable plants that naturally have low capacity to form adventitious roots or that loose this ability during maturation. AR development was shown to be controlled by a complex network of plant hormones crosstalk, in which auxin signaling plays a central role in each step of the process. Auxin application to the cuttings base is the common treatment to induce adventitious root formation, however, many recalcitrant plants do not make roots in response to such treatment.
Our work in this subject focuses on two areas: 1. The development on new rooting enhancers, in collaboration with Dr Roy Weinstain from the Tel-Aviv University. Our new compounds are tested for their ability to enhance adventitious root formation in recalcitrant plants such as Eucalyptus trees, Argan, Pecan root stocks, Guava, Protea species and more. 2. The other area includes the cell biology underlying adventitious root formation. In this area we address questions related to the contribution of cell wall modifications to differentiation of adventitious roots as well as different aspects of membrane trafficking, protein-protein interactions and post translational modifications affecting adventitious root formation, or instead callus formation.
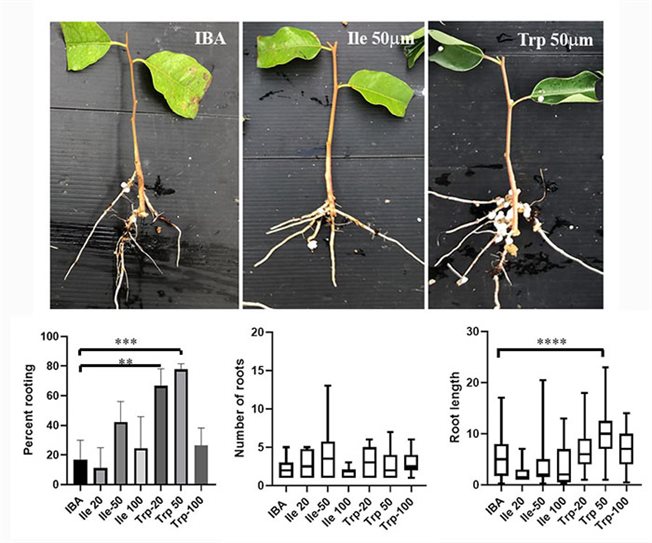
Rooting of cuttings from mature Eucalyptus grandis trees using conjugates as new rooting enhancers

Differentiated cells in callus formed at the cutting base of Avocado rootstock. A. Circular vessels. B. Cork. C-D. Amyloplasts in storage tissue, C-Bright filed, D- Polarized light. E. Rooted cutting with callus and root.
Partial list of publications (for the full list please see:)
https://scholar.google.com/citations?user=rqoVQqwAAAAJ&hl=iw
Bar-Sinai S, Belausov E, Dwivedi V, Sadot E. (2022). Collisions of Cortical Microtubules with Membrane Associated Myosin VIII Tail. Cells 11(1): 145.
Duman, Z., Hadas-Brandwein, G., Eliyahu, A., Belausov, E. Abu-Abied, M. Yeselson, Y., Faigenbaum, A., Lichter, A., Irihimovitch, V. and Sadot, E. (2020) Short de-etiolation increases the rooting of VC801 avocado rootstock. Plants (Special issue for Adventitious root formation in tree species). 9(11), 1481
Riov, J., Fox, H., Attias, R., Shklar, G., Farkash-Haim, L., Sitbon, R., Moshe, Y., Abu-Abied, M., Sadot, E., and David-Schwartz, R. (2020). Improved method for vegetative propagation of mature Pinus halepensis and its hybrids by cuttings. Isr. J. Plant Sci. 67: 5-15.
Duman, Z., Eliyahu, A., Abu-Abied, M., and Sadot, E. (2020). The contribution of cell wall remodeling and signaling to lateral organs formation. Isr. J. Plant Sci., 67:110-127.
Teper-Bamnolker, P., Danieli, R., Peled-Zehavi, H., Belausov, E., Abu-Abied, M., Avin-Wittenberg, T., Sadot, E., and Eshel, D. (2020) Vacuolar processing enzyme translocates to the vacuole through the autophagy pathway to induce programmed cell death. Autophagy. 19:1-15
Eliyahu A., Duman Z., Sherf S., Genin O., Cinnamon Y., Abu-Abied M, Weinstein R, Dag A, and Sadot E**. (2020) Vegetative propagation of elite Eucalyptus clones as food source for honeybees (Apis mellifera); adventitious roots versus callus formation. Israel J. Plant Sci. Special issue: Adventitious Root Formation; from the Lab to the Green house. Isr. J. Plant Sci. 67:83-97.
Sadot E. and Blancaflor E (2019) The actomyosin system in plant cell division: lessons learned from microscopy and pharmacology. In: The Cytoskeleton: Diverse roles in a plant's life. Eds. Baluska F. and Vaidurya. S. Springer
Abu-Abied M, Belausov E, Hagay S, Peremyslov V, Dolja V, Sadot E. Myosin XI-K is involved in root organogenesis, polar auxin transport, and cell division. J Exp Bot. 2018;69(12):2869-2881.
Chaimovitsh, D., Shechter, A., Abu-Abeid, M., Rubin, B., Sadot, E., and Dudai, N.(2016) Herbicidal Activity of Monoterpenes Is Associated with Disruption of Microtubules Functionality and Membrane Integrity. Weed Sci. 65(1):19-30.
Abu-Abied M, Mordehaev I, Sunil Kumar GB, Ophir R, Wasteneys GO, Sadot E. (2015) Analysis of Microtubule-Associated-Proteins during IBA-Mediated Adventitious Root Induction Reveals KATANIN Dependent and Independent Alterations of Expression Patterns. PLoS One. 2015 Dec 2;10(12).
Abu-Abied, M., Rogovoy (Stelmakh), O., Mordehaev, I., Grumberg, M. Elbaum, R., Wasteneys, G.O. and Sadot, E. (2015) Dissecting the contribution of microtubules behavior in adventitious root induction J Exp Bot. 66(9):2813-24.
Henn A, Sadot E. (2014) The unique enzymatic and mechanistic properties of plant myosins. Curr Opin Plant Biol. 2014 Dec;22:65-70.
Buchnik L, Abu-Abied M, Sadot E (2014) Role of plant myosins in motile organelles: is a direct interaction required? J Integr Plant Biol. 2015 Jan;57(1):23-30.
Abu-Abied M, Szwerdszarf D, Mordehaev I, Yaniv Y, Levinkron S, Rubinstein M, Riov J, Ophir R, Sadot E (2014) Gene expression profiling in juvenile and mature cuttings of Eucalyptus grandisreveals the importance of microtubule remodeling during adventitious root formation. BMC Genomics. 2014 Sep 30;15:826.
Levy A., Szwerdszarf D., Abu-Abied M., MordehaevI., Yaniv Y., Riov J., Arazi T., and Sadot E (2014) Profiling microRNAs in Eucalyptus grandis reveals no mutual relationship between alterations in miR156 and miR172 expression and adventitious root induction during development. BMC Genomics 25;15(1):524
Sadot E. (2013) Plant compounds acting on the cytoskeleton. Applied Plant Cell Biology; Cellular Tools and Approaches for Plant Biotechnology. Eds: Peter Nick and Zdenek Opatrný. Springer. Pp. 301-323.
Altshuler O., Abu-Abied M., Chaimovitsh D., Shechter A., Frucht H., Dudai N. and Sadot E. (2013) Enantioselective Effects of (+)- and (-)-Citronellal on Animal and Plant Microtubules. J. Nat. Prod. 76(9):1598-1604
Riov J., Szwerdszarf D., Abu-Abied M., Sadot E. (2013) The molecular mechanisms involved in adventitious root formation. Plant Roots: The Hidden Half, 5th edition, Eds. A. Eshel and T. Beeckman, Taylor and Francis, LLC.
Abu-Abied, M. Szwerdszarf, D. Mordehaev, I. Levy, A. Rogovoy (Stelmakh), O. Belausov, E. Yaniv, Y. Uliel, S. Katzenellenbogen, M. Riov, J. Ophir, R. and Sadot E. (2012) Microarray analysis revealed upregulation of nitrate reductase in juvenile cuttings of Eucalyptus grandis which correlated with increased nitric oxide production and adventitious root formation. Plant J. 71(5):787-99
Chaimovitsh D., Rogovoy (Stelmakh) O., Altshuler O., Belausov E., Abu-Abied M., Rubin B., Sadot E. and Dudai N. (2011) The relative effect of citral on mitotic microtubules in wheat roots and BY2 cells. Plant Biol. 14(2):354-364.
Avisar D., Abu-Abied M., Belausov E. and Sadot E. (2011) Myosin XIK is a major player in cytoplasm dynamics and is regulated by two amino acids in its tail. J. Exp. Bot. 63(1):241-9.
Halpert M, Abu-Abied M, Avisar D, Moskovitz Y, Altshuler O, Cohen A, Weissberg M, Riov J, Gottlieb H.E, Perl A and Sadot E (2011) Rac-dependent doubling of HeLa cell area and impairment of cell migration and cell cycle by compounds from Iris germanicaProtoplasma. 248(4):785-797.
Chaimovitsh D, Abu-Abied M, Belausov E, Rubin B, Dudai N, Sadot E.(2010) Microtubules are an intracellular target of the plant terpene citral. Plant J. 61(3):399-408.
Avisar, D., Abu-Abied, M., Belausov, E., Sadot, E., Hawes C. and Sparkes I.A. (2009) A comparative study on the involvement of 17 Arabidopsis myosin family members on the motility of Golgi and other organelles. Plant Physiol. 150(2) 700-709.
Golomb L, Abu-Abied M, Belausov E, Sadot E. (2008) Different subcellular localizations and functions of Arabidopsis myosin VIII.BMC Plant Biol. 2008 Jan 8;8:3.
Zaffryar S, Zimerman B, Abu-Abied M, Belausov E, Lurya G, Vainstein A, Kamenetsky R, and Sadot E. (2007) Developmental specific association of amyloplasts with microtubules in scale cells of Narcissus tazetta. rotoplasma. 230(3-4):153-63.
Abu-Abied, M., Golomb, L., Belausov, E., Huang, S., Geiger, B., Kam, Z., Staiger,C.J. and Sadot E. (2006) Identification of Plant Cytoskeleton-Interacting Proteins by Screening for Actin Stress Fiber Association in Mammalian Fibroblasts. Plant J. 48;367-379.
Yasuor, H., Abu-Abied, M., Belausov, E., Madmoni, A., Sadot, E., Riov, J. and Rubin, B. (2006) Glyphosate-induced antherindehiscence in cotton is partially temperature-dependent and involves cytoskeleton and secondary wall modifications and auxin accumulation. Plant Physiol. 141(4):1306-15.
Lahav M, Abu-Abied M, Belausov E, Schwartz A and Sadot E. (2004) Microtubules of guard cells are light sensitive. Plant Cell Physiol. 45(5) 573-582.